
Category: Clinical Trials
- ALK Experts
- ALK International
- ALK Positive News
- Clinical Trials
- Government
- Living with ALK+
- Nutrition
- Patient & Care Partner Stories
- Powered By Patients
- Recipes
- Research
-

Gilteritinib for the Treatment of ALK positive non-small cell lung cancer (NSCLC)
This trial is for patients with Stage 4 ALK positive NSCLC that have progressed on an earlier generation ALK TKI AND lorlatinib, and on any number of other standard-of-care or trial treatments. The trial drug is gilteritinib (aka Xospata), which was approved by the FDA in 2018 for a type of leukemia, but has been…
-

Nuvalent Receives U.S. FDA Breakthrough Therapy Designation for NVL-655
CAMBRIDGE, Mass., May 16, 2024 /PRNewswire/ — Nuvalent, Inc. (Nasdaq: NUVL), a clinical-stage biopharmaceutical company focused on creating precisely targeted therapies for clinically proven kinase targets in cancer, today announced that the U.S. Food and Drug Administration (FDA) has granted breakthrough therapy designation (BTD) to NVL-655 for the treatment of patients with locally advanced or…
-
The Global Landscape of Research: ALK-Positive Cancer Therapeutic Trials Worldwide
In the ever-evolving arena of ALK-positive cancer therapies, clinical trials offer patients not only cutting-edge interventions but also a chance to contribute to the advancement of ALK cancer treatment. In examining international ALK cancer trials, we uncover both challenges and opportunities for those seeking innovative treatments beyond conventional options. ALK Positive is the first foundation…
-

Unlocking Hope: Navigating your Path to ALK-Positive Lung Cancer Clinical Trials
Participating in a clinical trial can provide hope for those with ALK-positive lung cancer, offering a glimpse into the realm of cutting-edge treatments. Understanding eligibility criteria can be paramount for those seeking to participate in a clinical trial. Here we will highlight some of the opportunities and challenges of clinical trial eligibility
-

University of Michigan Biopsy and Organoid Drug Screening Experience
I’m writing this on the return flight from the University of Michigan, where I underwent a thoracic biopsy for three lymph nodes that have been lighting up in my recent PET scans. As I am the ALK Positive patient advocate on the Judith Tam ALK NSCLC Research Initiative Scientific Advisory Board, l thought I’d do…
-

Unlocking Hope: Breakthroughs in ALK-Positive Cancer Research for All Ages
The spectrum of ALK-positive cancer knows no age limits. Meet the faces of courage: the ordinary individuals with the ALK-positive mutation. This mutation, notorious for lung cancer in adults, defies
-

Oncolytic Virus Research Collaboration: Memgen Inc., UMichigan, and ALK Positive Inc.
The ALK Positive NSCLC Research Acceleration Committee (RAC) systematically reaches out to selected biotech companies with NSCLC treatments in development and/or trial. In February of 2022, RAC member Ray Hall
-

The ALK Positive Lung Cancer Registry and Survey Need You!!
What do patients that go only a short time before tumor progression have in common? What do patients that have many years before tumor progression have in common? What do
-

You Can Find a Clinical Trial with the Help of ALK Positive Inc.
At any juncture in treatment, those with ALK Positive cancers are faced with hard choices. Whether you are first starting treatment or you are changing treatment due to progression or
-

The New ALK Inhibitor: NVL-655 Trial Expands to Europe!
Who is this trial for: NVL-655 is a 4th generation, brain-penetrant, ALK TKI inhibitor created to overcome several limitations observed with currently available therapies. NVL-655 is designed to have activity in tumors that have developed resistance to first, second, and third-generation ALK inhibitors, including tumors with the solvent front G1202R mutation or compound mutations G1202R/L1196M…
-
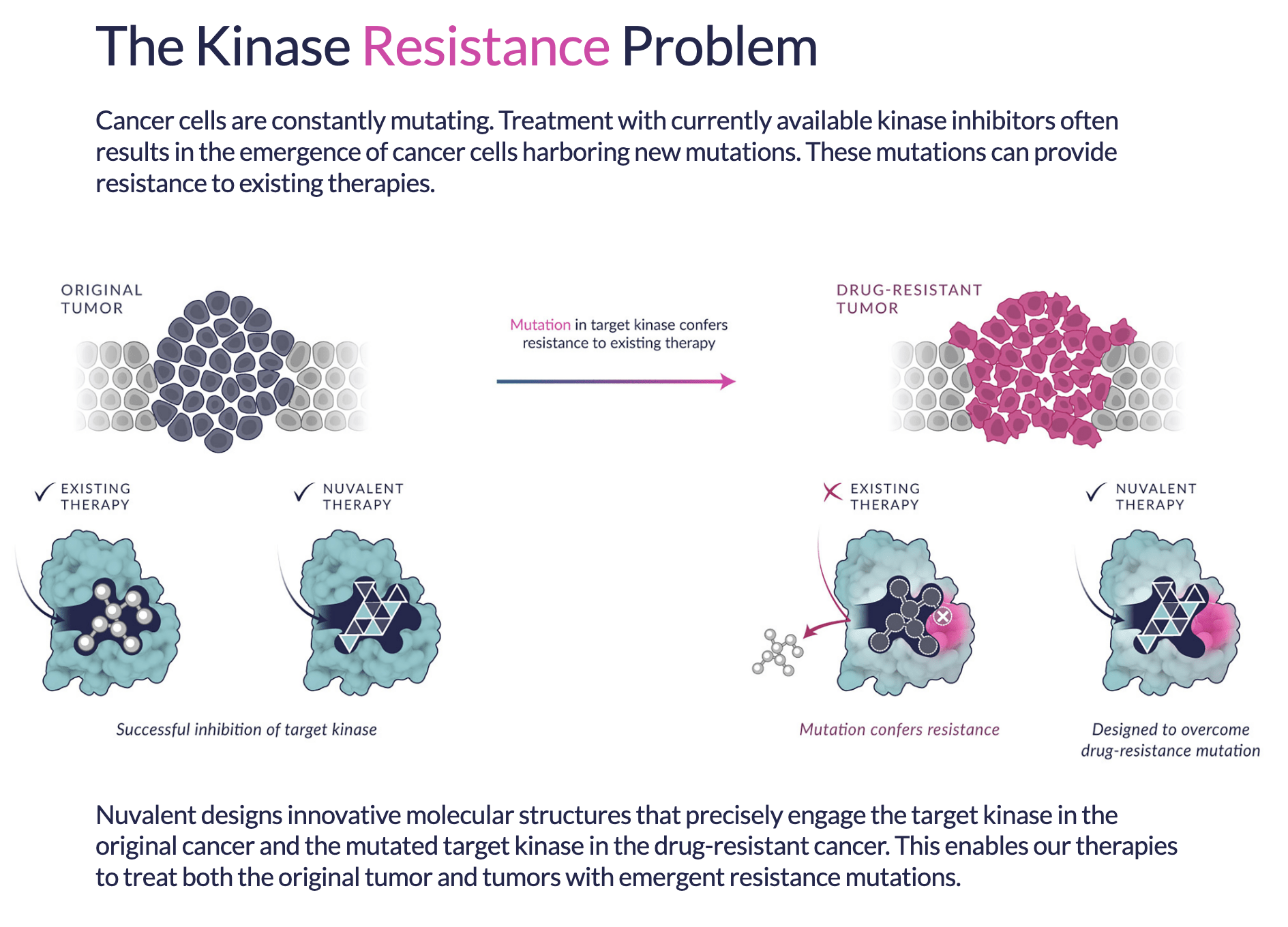
The New ALK Inhibitor: NVL-655 Expands Clinical Trial Sites
Who is this trial for: NVL-655 is a 4th generation, brain-penetrant, ALK TKI inhibitor created to overcome several limitations observed with currently available therapies. NVL-655 is designed to have activity in tumors that have developed resistance to first, second, and third-generation ALK inhibitors, including tumors with the solvent front G1202R mutation or compound mutations G1202R/L1196M…

