
Why Participate in a Clinical Trial?
Over 6,500 patients have enrolled in clinical trials over the last 12 years which have led to the approval of six tyrosine kinase inhibitors (TKIs), including Crizotinib (Xalkori), Ceritinib (Zykadia), Alectinib (Alecensa), Brigatinib (Alunbrig), Lorlatinib (Lorbrena), and Ensartinib (Ensacove) for the treatment of ALK-positive NSCLC.
Most people opt to participate in a clinical trial to have the opportunity to try a different treatment (or treatment combination) that may cure (totally eliminate) their cancer, or may give them a longer time before the cancer starts growing again, or may control cancer that is currently growing, and/or may provide them a better quality of life by reducing side effects of their cancer treatments.
Some people participate as social responsibility to advance medical care through research for future generations, and others opt to participate because they have a personal interest in science.
Participating in Clinical Trials
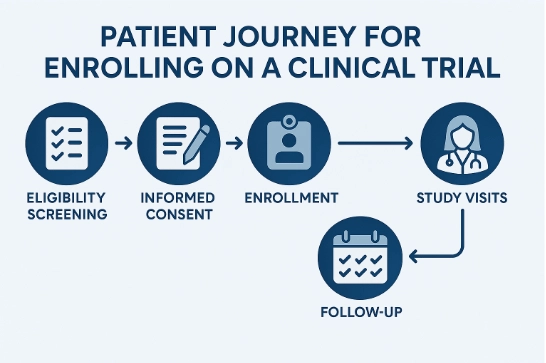
Patients participating in a clinical trial receive investigational treatments or combinations of treatments under the supervision of a physician and other research professionals. These treatments are typically developed by pharmaceutical and biotechnology companies who select qualified physicians, also known as investigators, to conduct clinical trials to determine the benefits of investigational treatments. When considering participation in a clinical trial, it is important to take the time you need to make an informed decision. Below are answers to frequently asked questions that many patients have about participating in a study.
Who Can Participate in a Clinical Trial?
All clinical trials have guidelines about who can participate. Before joining a clinical trial, a patient must “qualify” for the study. The factors that allow patients to qualify and participate in a clinical trial are called inclusion criteria and the factors that disallow patients from participating are called exclusion criteria. These criteria can include age, gender, the type and stage of the cancer, previous treatment history, and other medical conditions (e.g. heart disease).
It is important to note that inclusion and exclusion criteria are used to identify appropriate participants, promote participants’ safety, and ensure that researchers learn the information they need.

What Questions Should be Asked Before Choosing to Participate?
Patients who are considering participating in a clinical trial should review the consent form in detail and talk about it with their family, friends and personal physician. Patients should also review the credentials and experience of the staff and the facility involved in conducting the study.
Questions to ask the investigator and research staff:
● How long will the trial last?
● Where is the trial being conducted?
● What treatments will be used and how?
● What is the main purpose of the trial?
● How will patient safety be monitored?
● Are there any risks involved?
● What are the possible benefits?
● What are the alternative treatments besides the one being tested in the trial?
● Who is sponsoring the trial?
● Do I have to pay for any part of the trial?
● Is there reimbursement for costs I incur?
● How much time will I need to commit to participate?
● What happens if I am harmed by the trial or injured during the trial period?
● Can I opt to remain on this treatment, even after termination of the trial?
